0000000000000000000000000000000000
000000000000000000000000000000000
00000000000000000000000000000000
0000000000000000000000000000000
000000000000000000000000000000
00000000000000000000000000000
0000000000000000000000000000
000000000000000000000000000
00000000000000000000000000
0000000000000000000000000
000000000000000000000000
00000000000000000000000
0000000000000000000000
000000000000000000000
00000000000000000000
0000000000000000000
000000000000000000
00000000000000000
0000000000000000
000000000000000
00000000000000
0000000000000
000000000000
00000000000
0000000000
000000000
00000000
0000000
000000
00000
0000
000
00
0
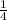 )
)
ответ: 6,72 л.
Дано:
m(Ca)=12 г.
M(Ca)=40 г/моль;
M(H2)=2 г/моль ;
Найти:
V(H2) - ?
Решение: Ca+H2SO4 = H2 + CaSO4
n(Ca)=m/M = 12 г. / 40 г/моль = 0,3 моль
n(Ca) : n (H2) = 1:1
n(H2) = 0,3 моль
V(H2)= n*Vm = 0,3 моль * 22,4 л/моль = 6,72 л.